Use coupon code MOISTURIZER with your purchase of $40 or more for a free†
Blemish Clear MoisturizerNutrition and Supplement Facts Label Changes: What’s New?

You may have noticed that the Nutrition Facts panel on food products and the Supplement Facts panel on dietary supplements look different. These changes have been made in response to new FDA labeling regulations, which went into effect on July 26, 2016. These changes affect several aspects of current labeling practices and serving sizes.
Supplement Facts and Nutrition Facts Panels – Key Changes:
The new regulations require changes in the units of measure for folic acid, and vitamins A, D, and E.

- Vitamin A will no longer be measured in IUs (International Units). Vitamin A will be measured in mcg RAE (micrograms Retinol Activity Equivalents) going forward. The RAE unit measure more accurately reflects the actual vitamin A activity of its many forms, including retinoids (e.g., retinol) and carotenoids (e.g., b-carotene).
- Vitamin D will now be measured in mcg (micrograms) instead of IUs; however, you may still see IUs in parentheses next to mcg on labels.
- Vitamin E will now be presented in mg (milligrams) RRR-a-Tocopherol instead of IUs. This change is being made to account for the difference in activity between naturally occurring and synthetic vitamin E. Vitamin E still only includes the a-tocopherol form and not the other tocopherol forms, nor the tocotrienols.
- Folic Acid will be measured as mcg DFEs (Dietary Folate Equivalents). This change addresses the difference in bioavailability between dietary folate and folic acid, which is the supplemental form.
The recommended Daily Values (DV) for most nutrients have changed.
- Many DVs have increased, including vitamin D, calcium, vitamin K, vitamin C, phosphorus, magnesium, manganese, and potassium. On these nutrients, you might see the %DV go down on many products.
- Other DVs have decreased. These include sodium, vitamin E, most B-complex vitamins, zinc, selenium, copper, chromium, and chloride. You may therefore notice that the %DV for these nutrients has gone up on many products.
- Only one nutrient, folate, had no change in recommended DV.
The new regulations affect dietary fiber declarations.
- The definition of dietary fiber has changed. The new definition focuses on reporting fiber that is considered beneficial to human health by FDA. Dietary Fiber in the new regulations includes non-digestible carbohydrates and lignins that are found naturally in plants, plus added fiber that FDA has determined is beneficial in humans. Fibers that meet this definition are included in the Dietary Fiber and Total Carbohydrate totals on the label.
- According to the new definition, ingredients such as psyllium, beta-glucan, guar gum, and pectin will still be considered to be dietary fiber. Other ingredients such as FOS (fructooligosaccharide) and inulin will not, even though studies have determined that these fibers can positively impact digestive function.
- The recommended DV for fiber has increased from 25 g/day to 27 g/day.
The Nutrition Facts panel on food products also looks different.
- The most noticeable change is the way Calories are displayed. Caloric content is in larger type and bold; “Serving size” is also in a larger, bold type.
A new line for Added Sugars helps the consumer determine how much of the sugar in the product has been added by the manufacturer and how much is naturally occurring.
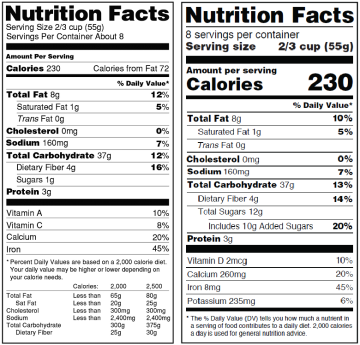
- This new line will show both number of grams and the percentage of the recommended Daily Value, which is the amount of sugar experts believe is reasonable with a 2,000 calorie daily diet.
- What may be confusing to consumers is the fact that even a pure sugar, such as beet sugar or fructose, can be considered an “Added Sugar” because it’s not being consumed in its natural state as a component of the natural source. So if you’re not eating a beet, then you’re not getting beet sugar from a natural source, which makes it an added sugar under the new guidelines.
Note that in addition to the DV (Daily Value) of the nutrients declared, actual amounts of these nutrients are now required.

NEW LABEL / WHAT'S DIFFERENT
- Servings: larger, bolder type
- Serving sizes updated
- Carolies: larger type
- Updated daily values
- New: added sugars
- Change in nutrients required
- Actual amounts declared
- New footnote











